About Siemens Opcenter Execution Medical Device & Diagnostics
Within the Siemens Opcenter MOM offering, Opcenter Execution is a portfolio of industry and process-specific Manufacturing Execution Systems (MES) designed to enable organizations to monitor, control and manage all the activities, resources and work-in-progress (WIP) on the factory floor and across their enterprise during the execution of production.
Siemens Opcenter Execution Medical Device and Diagnostics (formerly known as Camstar Medical Device Suite) is a leading manufacturing execution solution for error-proofing processes, paperless manufacturing and electronic Device History Record (eDHR) management. It helps you accelerate innovation, reduce costs and deliver the highest quality products possible while making compliance a by-product of excellent manufacturing processes. Opcenter Execution Medical Device and Diagnostics (MDD) enables you to build and validate quality within the manufacturing process itself, instead of testing quality coming out of the process at every step. From raw materials to finished goods, Opcenter Execution MDD delivers a complete and self-auditing eDHR solution.
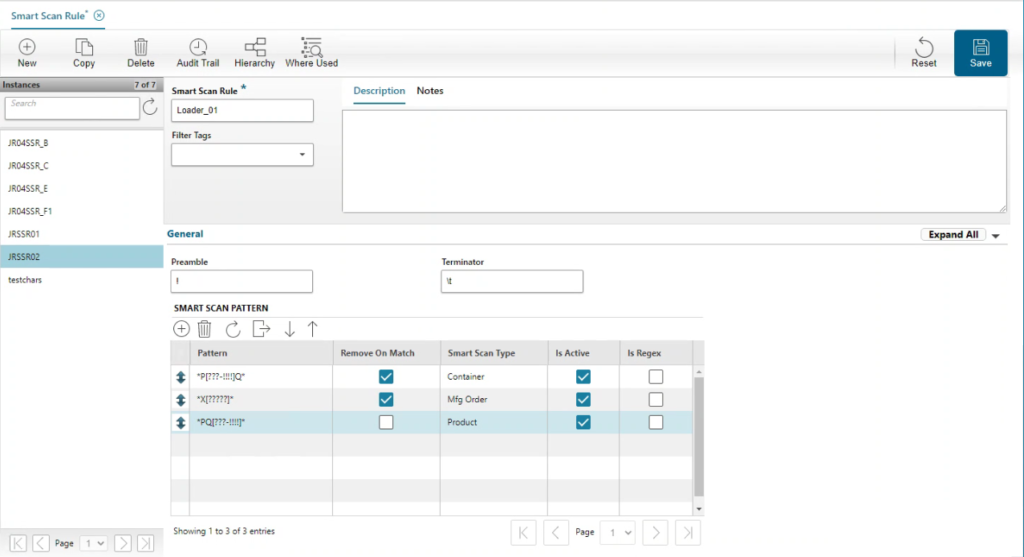
Opcenter Execution MDD automates and enforces complete control over the five “Ms” in Medical Device Manufacturing:
- Opcenter Execution MDD helps you manage Man: automatically checks training and certifications before allowing a user to work at an operation or inspection.
- Opcenter Execution MDD helps you manage Machine: ensures that only qualified equipment that is up-to-date on maintenance and calibration is used.
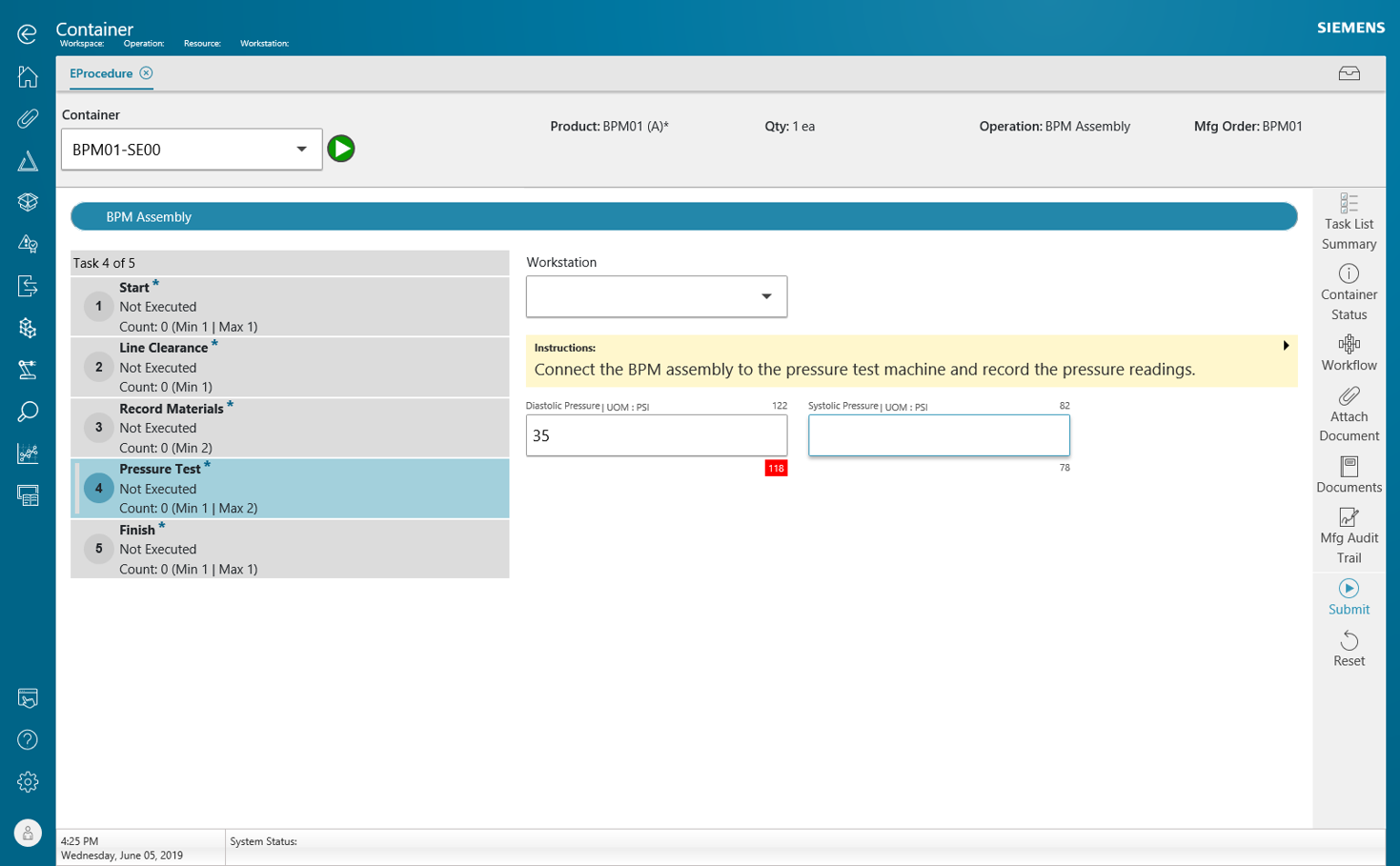
- Opcenter Execution MDD helps you manage Method: enforces process sequence, ensuring that all steps and data collection are complete and providing instant feedback to guarantee compliance.
- Opcenter Execution MDD helps you manage Material: ensures that operators use the right quantity of qualified materials, enforcing expiration dates, LIFO (Last In, First Out) and FIFO (First In, First Out) rules.
- Opcenter Execution MDD helps you manage Measurement: ensures that all required data is collected as compared to specification limits and automatically triggers rework or non-conformance workflows.
Opcenter Execution MDD was built integrating best practices in Medical Device Manufacturing, enabling faster out-of-the-box implementation. The software is highly functional and flexible, and provides an advanced and intuitive user experience. It also integrates fully with other enterprise systems, such as PLM, ERP, Production Planning & Scheduling and Manufacturing Intelligence (MI) platforms, helping you reap the benefits of true closed-loop manufacturing.